More complicated electronics will require the increased use of specialty solders. Specialty solders, such as indium alloys, offer advantages for gold soldering, step soldering and fatigue resistance.
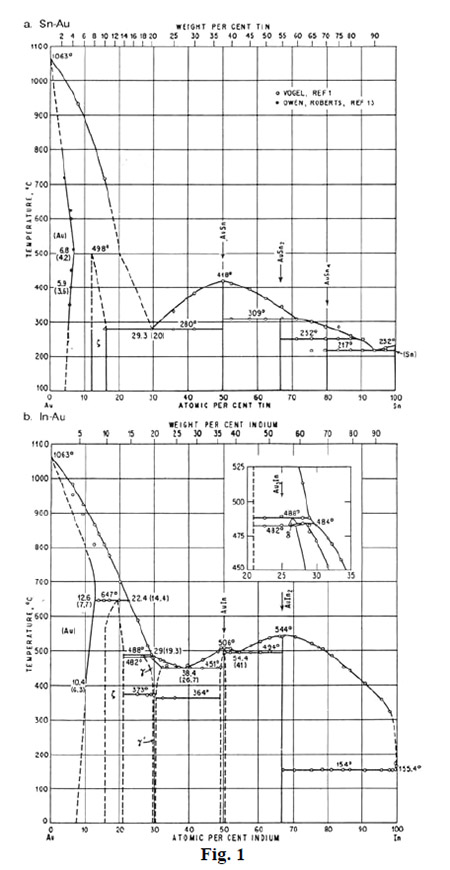
Indium/Lead Solders on Gold. Because they do not leach or dissolve gold as readily as tin/lead solders, indium/lead solders are recommended for soldering to gold. As seen in fig. 1, the phase diagrams of tin/gold shows that tin dissolves approximately 18 percent by weight of gold at soldering temperatures of 225-250°C.1 Under the same conditions the indium/gold phase diagram shows dissolution of only 2 to 4 percent of gold in indium. Thus, with a substantial reduction in the dissolution of gold, less intermetallics are formed and improved solder flow over the gold surface occurs.
Tin/lead soldering of gold is possible if the layer of gold is sufficiently thin (1.27 µm (50 µin) or less) to allow dipping in a pre-solder to remove the gold. However, if this does not occur then there is a high probability of solder joint failure. Immediately after soldering, a neat solder joint will be observed and it will pass electrical test. However, over time the tin will dissolve the gold by solid state diffusion and leave an opening in the electrical contact.
For the above reasons, whenever possible an indium/lead solder should be used on a gold alloy. If indium/lead cannot be used due to either environmental reasons or temperature conditions, it is recommended to use a gold/tin alloy. The two gold/tin alloys that are commonly used are the Sn90/Au80 217°C eutectic and the Au80/Sn20 280°C eutectic.
Indium/Copper Intermetallics. Some concern regarding indium intermetallic formation with copper exists due to previous studies.2 However, what should be noted is that this testing was performed at 150°C, which is a condition that is suitable for any soft solder including tin/lead. More pertinent tests on indium/tin and indium/lead have since been run at 80°C and do not show intermetallic formation. At 125°C, some intermetallics are formed, but are comparable to the amount formed by tin/lead at the same temperature. As seen in the bright areas of fig. 2, little evidence of the indium/copper intermetallic formation is seen in the EDAX spectra of indium/tin with a copper coupon.3 A barrier metal such as nickel should be used if higher temperatures are needed.

Thermal Fatigue Resistance of Solder Alloys. Indium/lead exhibits good thermal cyclic fatigue resistance because the solder does not crack during thermal cycling from -55° to 125°C. It is best to use indium/lead solders on very rigid structures such as ceramic-to-metal or ceramic-to-ceramic.
In addition, avoid high stress solder conditions where there is a drastic mismatch in the thermal expansion of materials. Do not solder rigid structures together using tin/lead. In a rigid structure, the tin/lead has all the forces exerted on it and will crack and again leave an opening in the electrical connection. In contrast, tin/lead solders are very strong, exhibit excellent wetting but have poorer fatigue resistance. They tend to crack after long-cycling periods with rigid structures such as ceramic-to-metal or ceramic-to-ceramic.
The use of a high tensile solder, such as tin/silver, is recommended on flexible circuits. Tin/silver and tin/antimony/silver exhibit very high joint strength and put the stresses back in the flexible part of the board, thus eliminating any solder fatigue.
 Indium Alloy Corrosion. Indium, indium/tin and indium/lead alloys are easily corroded by halides in the presence of moisture. If indium alloys are going to be used in this type of environment, they should either be hermetically sealed or conformally coated to prevent corrosion.
Indium Alloy Corrosion. Indium, indium/tin and indium/lead alloys are easily corroded by halides in the presence of moisture. If indium alloys are going to be used in this type of environment, they should either be hermetically sealed or conformally coated to prevent corrosion.
Indium oxide reacts with chlorine to form indium chloride which the reacts with carbon dioxide to form indium carbonate which in the presence of water forms indium hydroxide. To mitigate corrosion, samples using 85/80 indium/lead and other indium alloys should be conformal-coated for temperature testing in high humidity. Fig. 3 shows some corroded joints.
Solder Joint Appearance. Bright shiny joints are commonly sought during inspection. However, this requirement actually can cause problems in the long-term life of components. To satisfy the requirement for bright shiny joints, manufacturers will sometimes touch up the solder joint by reflow. Reheating indium/lead and, to a lesser degree, tin/lead, will lead to the formation of additional surface oxides which result in further corrosion, and more base metals are dissolved. The result is a very highly contaminated joint that could have a limited life. An untouched up joint with only 80 percent coverage would have a lower failure rate. Full fillet formation with bright shiny joints is definitely desirable; however, methods of achieving these must be controlled. If proper incoming solderability testing is performed and solder materials are tightly controlled, these conditions can be met. If touch-up is necessary, reflow the joint once at the correct temperature and time.
Solder joints using indium/lead eutectics are dull in appearance compared to shiny tin/lead alloys. Slight differences in the composition of tin/lead eutectics will also yield different appearances. For example, Sn62 (62% tin, 36% lead, and 2% silver) tends to be much grainier and frostier looking than Sn63 (63% tin and 36 lead) because of the silver content. When using specialty alloys, visual standards should be established and implemented in quality assurance to aid in the discernment of good joints; i.e., a standard joint should be fabricated and used as a control.
Soldered Structural Joints. Soft solders such as tin/lead or indium/lead should not be used for structural joints. Indium/lead eutectics are soft solders and strictly fillers for electrical contacts. Use a brazing (or hard) solder if structural strength is required. Normally, solder should not be used for any type of structural joint.
Solder Purity. Optimized solderability can be achieved by specifying tighter impurity levels on indium/lead solders. Bismuth, cadmium, and antimony levels that exceed 0.08 percent by weight tend to inhibit flow. If impurities are excessively high they will change the melting point of the alloy. Impurity levels in IPC-JSTD-006 are adequate for indium/lead alloys with the exception of bismuth. The purer metals wets slightly better, especially in the surface mount applications. Low temperature solders (i.e., below the tin/lead eutectic) have a specification written through ASTM. Procurement of some low temperature alloys can be received by implementing this specification.
Step Soldering. Because the indium/lead system is a solid solution it can be used easily for step solders. Since there is no eutectic in the system, the temperature range can be changed very slightly by changing the indium:lead ratios.
Summary. More complicated electronics will require the increased use of specialty solders. Specialty solders, such as indium alloys, offer advantages of reduced intermetallic formation, gold soldering, and thermal fatigue resistance.
1 “Binary Alloy Phase Diagrams,” American Society of Metals, Metals Park, OH(1986) Volume 1.
2 Ron Kossowsky, “ Corrosion of Indium Based Solders,” Westinghouse research & Development Center, Pittsburgh, PA 15235; R.C. Pearson and L.T. Christovich, Westinghouse Systems Development Division, Baltimore, MD 20205.
3 K.F. Seelig, D.M. Sklarski, L. Johnson, and J. Startell, “The Advantages of Low Melting Temperature Solder when Applied to Plated-Through Hole Technology,” Nepcon East 1987



