Comparing Properties of CASTIN™, BABBITT & Alternative Lead-Free Alloys
Abstract. This paper compares three commercial lead-free solders; CASTIN®, Babbitt and SAC-I and their modified compositions in terms of mechanical and soldering properties. Solders tested are six alloys with different compositions in the range of (major elements): Sn/Cu(0.7-4)/Ag(0-4)/Sb(0.5-8)/Bi(0-3)/Ni(0-0.15). Effects of alloying elements on the thermal behavior (melting and solidification), wetting force, contact angle, spreading, tensile and hardness properties are studied. Based on preliminary results an alloy modification is made and its properties are compared and discussed against the commercial creep resistant solder alloy, i.e. SAC-I.
Key words:
Lead free solder, wetting, mechanical properties, thermal behavior
Introduction. Solder plays an important role in the assembly and interconnection of electronic materials. Traditionally, Sn-Pb alloy has been the major solder alloy for electronic owing to its low cost and performance. The eutectic 63Sn-37Pb solder has preferred soldering characteristics and reliability for microelectronic assembly. However, the inherent toxicity of lead and associated environmental and public health concerns is driving the microelectronic industries to seek suitable solder alloys to replace traditional Sn-Pb solder alloy [1-4]. Many lead-free solder alloys have been developed and investigated as alternatives for the conventional eutectic Sn-Pb. Among the lead-free alternatives, Sn-Ag-Cu (SAC) alloys have received the most attention and regarded as the de facto replacement for Sn-Pb solders.
 The increasing demand for high reliability electronics in the automobile industry is driving the electronics industry to seek for solders that can tolerate temperature fluctuations between cold and hot extremes. The traditional lead-free SAC alloys have poor creep resistance in harsh service environments [5]. During aging, SAC alloys undergo substantial microstructure evolution which manifests itself as a change in mechanical behavior as shown in Fig.1. Such a degradation of mechanical strength represents a significant problem for solder joint reliability. The addition of alloying elements such as bismuth (Bi), antimony (Sb) and nickel (Ni) can improve the mechanical performance of solder alloys. Bismuth dissolves in the tin matrix and improves strength and creep resistance due to the solid solution hardening effect [6]. It has also been reported that antimony in solution (in Sn) has only a minor effect on the creep resistance [7], however, alloys with higher concentrations of antimony show a uniform distribution of Sn/Sb precipitates which provide a profound strengthening effect reducing the creep rate of the alloy [8]. Nickel is marginally soluble in tin and forms an intermetallic compound phase that can increase strength due to precipitation hardening and also reduces the sliding of the grain boundaries which improves creep performance [9]. In this paper, therefore, we study mechanical and soldering properties of an alloy developed for high operating temperature named SAC-I and compare it with commercial lead-free alloys; CASTIN® and Babbitt and their modifications.
The increasing demand for high reliability electronics in the automobile industry is driving the electronics industry to seek for solders that can tolerate temperature fluctuations between cold and hot extremes. The traditional lead-free SAC alloys have poor creep resistance in harsh service environments [5]. During aging, SAC alloys undergo substantial microstructure evolution which manifests itself as a change in mechanical behavior as shown in Fig.1. Such a degradation of mechanical strength represents a significant problem for solder joint reliability. The addition of alloying elements such as bismuth (Bi), antimony (Sb) and nickel (Ni) can improve the mechanical performance of solder alloys. Bismuth dissolves in the tin matrix and improves strength and creep resistance due to the solid solution hardening effect [6]. It has also been reported that antimony in solution (in Sn) has only a minor effect on the creep resistance [7], however, alloys with higher concentrations of antimony show a uniform distribution of Sn/Sb precipitates which provide a profound strengthening effect reducing the creep rate of the alloy [8]. Nickel is marginally soluble in tin and forms an intermetallic compound phase that can increase strength due to precipitation hardening and also reduces the sliding of the grain boundaries which improves creep performance [9]. In this paper, therefore, we study mechanical and soldering properties of an alloy developed for high operating temperature named SAC-I and compare it with commercial lead-free alloys; CASTIN® and Babbitt and their modifications.
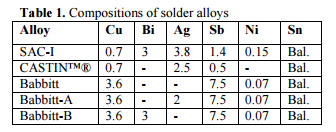 Experimental. Five alloys were prepared in an electric pot with graphite crucible. The composition of solder alloys was analyzed by arc emission spectroscopy is shown in Table 1. The molten alloys were cast into flat dog-bone shape in a stainless steel mold for tensile testing. The tensile specimens with the dimensions shown in Fig.2 were annealed at 125°C for 96 hr. to remove any residual stress or inhomogeneity induced during the sample preparation. Tensile tests were performed at room temperature with a constant strain rate of 10-3 s-1. For each alloy at least five specimens were tested to ensure reproducibility of the tensile test results. Hardness of all the specimens was also measured by Vickers microhardness testing machine with 25 gf load and 15s impression time.
Experimental. Five alloys were prepared in an electric pot with graphite crucible. The composition of solder alloys was analyzed by arc emission spectroscopy is shown in Table 1. The molten alloys were cast into flat dog-bone shape in a stainless steel mold for tensile testing. The tensile specimens with the dimensions shown in Fig.2 were annealed at 125°C for 96 hr. to remove any residual stress or inhomogeneity induced during the sample preparation. Tensile tests were performed at room temperature with a constant strain rate of 10-3 s-1. For each alloy at least five specimens were tested to ensure reproducibility of the tensile test results. Hardness of all the specimens was also measured by Vickers microhardness testing machine with 25 gf load and 15s impression time.
Thermal behavior of the alloys was investigated with differential scanning calorimetry (DSC). A piece of 20-25mg alloy was heated in an aluminum cell at heating rate of 10°C/min up to 300°C under nitrogen atmosphere followed by cooling to room temperature (ROC) at -10°C/min. Each alloy was tested twice and the reading was based on the second run.
Wettability is a critical factor when evaluating the performance of a solder alloy. The wetting process is dynamic during soldering and the solid/liquid interfacial tension between solder and substrate varies as the wetting proceeds. Wettability can be quantified with wetting balance technique that measures wetting force between molten solder and solid substrate versus wetting time. Wettability of the alloys was therefore quantified with wetting balance technique. A copper plate coupon (25mm×6mm×0.5mm) used as the substrate was immersed in a bath of liquid solder alloy and the wetting force was measured with time. A commercial alcohol based no clean flux (AIM NC 265) was used to clean the substrate surface. Each test was repeated three times and the average value was reported.
 Wetting contact angle (Fig. 3) was measured for selected alloys by means of Drop Shape Analyzer (DSA 100) Kruss machine. Using a commercial water based no clean liquid flux (AIM NC 275) 50±2mg solder alloy was melted on a smooth copper plate at 260°C and the contact angle was measured in the liquid state. The results are average of five testing measurements.
Wetting contact angle (Fig. 3) was measured for selected alloys by means of Drop Shape Analyzer (DSA 100) Kruss machine. Using a commercial water based no clean liquid flux (AIM NC 275) 50±2mg solder alloy was melted on a smooth copper plate at 260°C and the contact angle was measured in the liquid state. The results are average of five testing measurements.
The specimens for microstructural observation were successively ground on progressively finer papers from 360 grit to 800 grit. Polishing was performed using 1 μm diamond suspension followed by 0.05 μm alumina suspension followed by washing with alcohol. The specimens were subsequently etched by immersing in a solution of 2% (by volume) hydrochloric acid (HCl) and 5% nitric acid (HNO3) in methanol for about 5-15 s. Right after etching the specimens were washed with alcohol. Microstructures of selected alloys were observed with optical microscope.

Results and Discussion. DSC heating curves (heat flow versus temperature) are shown in Fig. 4. The valleys stand for intense endothermic phase transitions, i.e. melting. Except for Babbitt-A that shows two peaks (valleys) other alloys have one endothermic melting peak. For onset melting temperature (solidus) determination linear extrapolation method is utilized and the results are depicted in Fig. 5. Babbitt melts at a high temperature, 237°C, but adding Bi or Ag to Babbitt reduces the melting temperature. It is interesting to note that 2% Ag has a more significant effect on reducing the melting point (i.e. 13°C) as compared to the addition of 3% Bi (i.e. 6°C). In other words, Ag is a much more efficient element to lower the melting temperature of Babbitt alloy as compared to Bi.
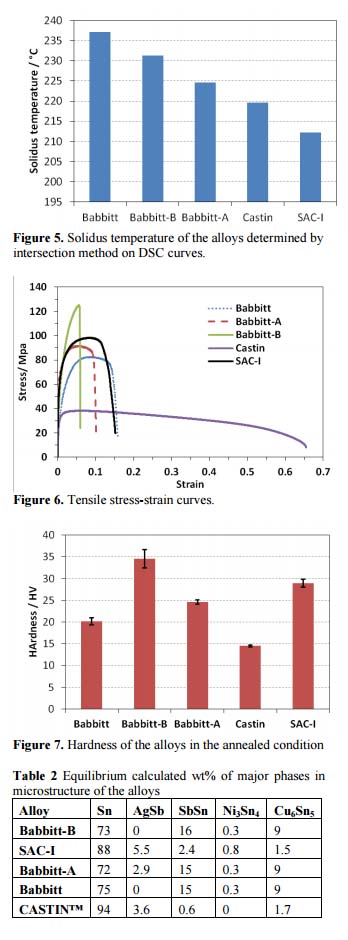 Tensile stress-strain curves of the alloys in fig. 6 show that SAC-I has a higher strength compared to Babbitt, but addition of Bi increases the strength of Babbitt by solid solution strengthening mechanism and at the same time makes it less ductile. Ag has little effect on tensile behavior of Babbitt. Comparatively, CASTIN® is the weakest but the most ductile alloy in Fig. 5. Furthermore, consistent with tensile test results, hardness data (Fig. 7) also demonstrates Babbitt-B as the hardest alloy and CASTIN® as the softest alloy in this study.
Tensile stress-strain curves of the alloys in fig. 6 show that SAC-I has a higher strength compared to Babbitt, but addition of Bi increases the strength of Babbitt by solid solution strengthening mechanism and at the same time makes it less ductile. Ag has little effect on tensile behavior of Babbitt. Comparatively, CASTIN® is the weakest but the most ductile alloy in Fig. 5. Furthermore, consistent with tensile test results, hardness data (Fig. 7) also demonstrates Babbitt-B as the hardest alloy and CASTIN® as the softest alloy in this study.
In order to better understand effect of alloying elements on microstructure and mechanical behavior of the alloys, phase equilibria calculation was carried out by means of the FactSage™ thermodynamic software [10]. The calculated weight percent of major phases present in each alloy is illustrated in Table 2. The equilibrium microstructure of Babbitt alloys consists of large amount of precipitates Sb/Sn and Cu6Sn5 whereas SAC-I has much lower precipitate phases. This leads to a conclusion that the high strength of SAC-I which is comparable to Babbitt alloys is attributed to solid solution strengthening of Bi and perhaps precipitation hardening effect of Ni elements. CASTIN® is a relatively soft alloy because does not contain large amount of solid solution and particle strengtheners.
Optical microstructures of the hardest and softest alloys, i.e. Babbitt-B and CASTIN®, as well as SAC-I are shown in Fig. 8. It is illustrated that CASTIN® is a eutectic type dendritic microstructure, whereas Babbitt-B comprises Sn-rich matrix (dark color) and large SnSb (β phase) and small Cu6Sn5 particles (η phase). SAC-I microstructure shows similar but lower amount of precipitates as that of Babbitt-B. The presence of these particles as well as the dissolution of bismuth in tin increases the strength of the alloy through solid solution and precipitation strengthening mechanisms. This is reflected in the mechanical behavior of the alloys presented in Fig.6 and Fig.7.

The wetting balance test results shown in Fig. 9 depicts that bismuth and silver additions similarly improve wettability of Babbitt by increasing wetting force and reducing wetting time demonstrated with the shifting wetting curve towards top left of Babbitt curve. Comparatively, SAC-I and CASTIN® have better wetting behavior with a higher wetting force and shorter wetting time. Moreover, the wetting appearance of the solders demonstrated in Fig. 10 ranks CASTIN® as the best solder with a smooth surface whereas other alloys’ surfaces are fairly rough.
Considering melting, wetting and mechanical behavior of the alloys studied, CASTIN™ and SAC-I are better choices for further evaluation. One drawback of SAC-I alloy, however, is its poor formability due to the high content of strengthening alloying elements. It is too hard to draw and make solid and cored wire. CASTIN™ formability is superior to SAC-I and can be drawn easily, however, it has lower mechanical and creep resistance. Thus, an attempt was made to improve the mechanical performance of CASTIN™ while maintaining its formability properties. Fig. 11 illustrates that a modified composition CASTIN™ alloy has similar melting behavior and tensile strength with much better formability as compared to SAC-I. This new alloy also demonstrates slightly better wetting behavior with smooth and continuous wetting balance curve as compared to that of SAC-I (Fig. 12). Wetting contact angle of the two alloys are comparable as illustrated in Fig. 13.




Summary. Soldering and mechanical properties of the following commercial lead-free solder alloys have been studied. The alloys investigated are:
Babbitt (3.6Cu/7.5Sb/0.07Ni),
Babbitt-A (Babbitt+2Ag),
Babbitt-B (Babbitt+3Bi),
SAC-I (0.7Cu/3Bi/3.8Ag/1.4Sb/0.15Ni) and
CASTIN™ (0.7Cu/2.5Ag/0.5Sb) (balance is Sn).
The following conclusions are drawn from this work:
- Melting temperature (Tm) of Babbitt is too high (237°C), but addition of Bi and in particular Ag reduce Tm. SAC-I shows the lowest melting temperature (212°C).
- Babbitt alloys and SAC-I show high strength and hardness with low ductility. Babbitt-B is very brittle while CASTIN™ is the most deformable alloys studied.
- Wetting behavior of SAC-I and CASTIN™ are equally good wheres Babbitt alloys show poor wettability.
Based on the soldering and mechanical data obtained from the mentioned alloys a new alloy named “modified CASTIN™" has been developed that shows comparable melting temperature, wettability and strength and better ductility with respect to SAC-I alloy. Further studies are required to investigate creep properties and solder joint reliability of the alloy.
REFERENCES
[1] M.M. Schwartz, ASM handbook: vol. 6, Welding, Brazing and Soldering, editors: D.L. Olson, T.A. Siewert, S. Liu, G.R. Edwards, ASM Int.1993, 126-137.
[2] L. Zhang, C.-W. He, Y.-H Guo, J.-G Han, Y.-W Zhang, X.-Y Wang, Microelectronics Reliability 52 (2012) 559-578.
[3] G. Zeng, S. Xue, L. Zhang, L. Gao, J Mater Sci : Mater Electron (2011) 22 : 565-578.
[4] M. Abtew, G. Selvaduray, Mater Sci Eng 27 (2000) 95-141.
[5] A.Z. Miric, SMTA Int. Conf., Orlando, FL, Oct. 2010.
[6] R. Mahmudi, A. Maraghi, Mater. Sci. Eng. A 599 (2014) 180-185.
[7] R.J. McCabe, M.E.Fine, Metall.Mater.Trans.A33(2002)1531–1539.
[8] R. Mahmudi, A.R.Geranmayeh, A.RezaeeBazzaz, Mater.Sci.Eng.A448 (2007) 287-293.
[9] H.-J. Albrecht, P. Frühauf, K. Wilke, SMT Int., 2009.
[10] C.W. Bale, P. Chartrand, S.A. Degterov, G. Eriksson, K. Hack, R. Ben Mahfoud, J. Melancon, A.D. Pelton, S. Petersen, CALPHAD 26 (2002) 189-228.



